Potential of Ficus Racemosa Bark: An immunomodulatory agent Potential of Ficus Racemosa Bark: An immunomodulatory agent
- by 1Rishikesh K. Jagzap, 2Sunil A. Nirmal* & 3Sagar K. Kadam
a) Abstract b) Introduction
c) Materials and Methods
d) Results e) Discussion
f ) References |
|
 |
|
Introduction: Traditionally, Ficus racemosa , the plant is used for various health problems, diseases and to improve immunity. Hence, we planned to validate its use as immunomodulatory, scientifically.
Materials & Methods: Petroleum ether, ethanol and aqueous extracts of bark of F. racemosa (50 and 100 mg/kg i.p.) was screened for immunomodulatory activity by delayed type hypersensitivity (DTH), neutrophil adhesion test and cyclophosphamide-induced neutropenia to assess the effect on immunity in Swiss albino mice. Preliminary phytochemical tests were done for active extract. Aqueous extract showed potentiation of DTH response in terms of significant increase in the mean difference in foot-pad thickness.
Results: Aqueous extract significantly increased neutrophil adhesion to nylon fibers by 45.75%, and 44.57% at 50 and 100 mg/kg dose, respectively. Aqueous extract significantly opposed reduction in TLC and neutrophil induced by cyclophosphamide. Percentage reduction in TLC and neutrophil by aqueous extract was 33.85%, 32.23% and 15.60 %, 13.20 % at 50 and 100 mg/kg dose respectively. Petroleum ether and ethanolic extract was found to non-significant compared to aqueous extract in above tests.
Conclusion: It is concluded that aqueous extract is a promising drug with immunostimulant properties. The extract was found to be most effective at low dose (50 mg/kg, i.p.), whereas, high dose (100 mg/kg, i.p.) of F. racemosa was moderately effective in modulating immune system, thus have great potential as a source for natural health products.
Keywords: Ficus racemosa, Neutropenia, Delayed type hypersensitivity, Immunomodulant adhesion. .
|
Back to Top  |
|
 |
 |
|
Indian traditional systems of medicines like Siddha and Ayurveda have
suggested increasing the body’s natural resistance to disease. Many clinical disorders are associated with the immune system. Suppression of the immune system is required in the management and treatment of inflammation and allergic diseases, while stimulation is highly desirable for the treatment of HIV, immunodeficiency and infectious diseases.1 The body's ability to defend itself against the infectious organisms in our environment is caused by the action of remarkable organization of interrelated cells and their biologic mediators, collectively known as the immune system.2 Ayurveda, the Indian traditional system of medicine, lays emphasis on promotion of health concept of strengthening host defenses against different diseases.3 F. racemosa Linn (Moraceae) is a moderate to large size spreading laticiferous tree without much prominent aerial roots. Leaves are dark green, ovate or elliptical. Bark is grayish green having soft surface while inner surface is light brown.
Fracture is fibrous. Plant is used in dysentery, pectoral complaints, diabetes, applied in mumps, other inflammatory glandular enlargements and hydrophobia.4,5 Stem bark showed the presence of two leucoanthocyanins: leucocyanidin-3-O-α-glucopyranoside, leucopelarogonidin - 3 - O - α- L - rhamnopyranoside, β-sitosterol, unidentified long chain ketone, ceryl behenate, lupeol, its acetate, and α-amyrin acetate.6,7 From trunk bark, lupeol, β-sitosterol, and stigmasterol were isolated.8,9 Glauanol, hentriacontane, β- sitosterol, gluanol acetate, glucose, tiglic acid, esters of taraxasterol, lupeol acetate, friedelin, higher hydrocarbons and other phytosterol are isolated from fruit.10,11 New tetracyclic triterpene glauanol acetate which is characterized as 13α, 14β, 17 βH, 20 αH-lanosta- 8, 22-diene-3 β-acetate and racemosic acid was isolated from the leaves.12 An unusual thermo stable aspartic protease was isolated from latex of the plant13.
Stem bark of the plant posse’s hypoglycemic activity and β- sistosterol was isolated from the stem bark14. Plant has antidiuretic15, wound healing16, hepatoprotective17, and analgestic activities18. Though a number of medicinal plants are available and traditionally used to treat variety of disease conditions but much of them remain to be explored scientifically for their immunomodulatory effects. Since natural products, especially Indian medicinal plants, are the potential source of immunomodulatory compounds19, the present study was performed to evaluate F. racemosa bark for its possible immunomodulatory activity in experimental animals. |
Back to Top  |
|
 |
 |
|
|
Plant material: Fresh sample of bark of F. racemosa was collected from Ahmednagar district, Maharashtra and authenticated by Dr. T. Chakraborty, Scientist D, Botanical Survey of India, Pune. A voucher specimen number JAGRISFICRA09 is being maintained.
Preparation of extract:
Shade dried bark of F. racemosa (180 g) was pulverized in form of moderately coarse powder (40 mesh size) and extracted successively in Soxhlet extractor using petroleum ether and ethanol to obtain respective extracts (2.8% and 8.4%, respectively). Marc left was refluxed with water for 6 hours to obtain aqueous extract (10.8%).
Drugs and chemicals:
Cyclophosphamide (cyc-negative control) was obtained from Endoxan®, Mumbai, Ethanol and EDTA solution AR from Merck, Mumbai, and levamisole (standard) was obtained from Bluck Pharma, Kolhapur.
Sheep RBC (SRBC):
Sheep blood was collected from local slaughterhouse in sterile 0.49% EDTA solution in 1:1 proportion of EDTA in saline (freshly prepared). Blood was kept in the refrigerator and processed for the preparation of sheep RBC by centrifugation at 2000 rpm for 10 min and washing with physiological saline for 4-5 times.
Animals:
Swiss albino mice of either sex weighing 20-25 gm were housed and were maintained at standard laboratory conditions. All mice were fed with synthetic pellet diet (Amrut laboratory animal feed, Sangli, Maharashtra) and clean tap water ad libitum. Mice were maintained at 22 ±1°C with 60% relative humidity, and kept under 12 h light:dark cycle. The animals were allowed to acclimatize to laboratory conditions prior to experimentation. The experimental protocol was approved by institutional animal ethical committee (Approval No. CPCSEA/C/448/10-11-/05).
Immunomodulating study:
Delayed type hypersensitivity (DTH)
The activity was performed as described by Bafna & Mishra (2004)20 and Devhare et al. (2011)21 . Animals were divided into nine groups consisting of six animals each. Control group received vehicle (1 ml/kg distilled water, p.o.) from first to seventh day of the study. Second groups received levamisole (50 mg/kg, i.p.) from first to seventh day of the study. Third group received cyclophosphamide as negative control (50 mg/kg, i.p.) from fourth to sixth days of the study. Fourth to ninth groups received petroleum ether, ethanol, and aqueous extracts (50 and 100 mg/kg, p.o., each) respectively from first to seventh days. On 0th day of the study, all the groups were immunized with SRBC (20%, s.c.) in normal saline. On 7th day all the animals from all groups were challenged with 0.05 ml of 1% SRBC in sub plantar region of right hind paw. Foot pad edema in mice was used for detection of cellular immune response. On 7th day, the initial thickness of right hind footpad was measured using digital vernier caliper and 24 hrs after the injection of 0.05 ml of 1% SRBC. Foot pad reaction was assessed after 24 hrs i.e. on 8th and 9th day, in terms of increase in the thickness of footpad due to edema caused as a result of hypersensitivity reaction, with the help of a digital vernier caliper22. The footpad reaction was expressed as the difference in the thickness (mm) between the pre and post right foot pad injected with SRBC.
Neutrophil adhesion test
:
Neutrophil adhesion test was performed as per the method described by Fulzele et al. (2002)23 and Devhare et al. (2011)21 . The animals were divided into eight groups consisting six animals each. Control group received vehicle (1 ml/kg distilled water, p.o.) from first to fourteenth days. Second groups received levamisole (50 mg/kg, p.o.) from first to fourteenth days. Third to eight groups received petroleum ether, ethanol, and aqueous extract (50 and 100 mg/kg, p.o., each), respectively from first to fourteen days. On the 14th day of the treatment, blood samples from all the groups were collected by puncturing retroorbital plexus under mild ether anesthesia. Blood was collected in vials pre-treated by disodium EDTA and analyzed for total leukocyte count (TLC) and differential leukocyte count (DLC) by fixing blood smears and staining with Leishman’s stain. After initial counts, blood samples were incubated with nylon fiber (80 mg/ml of blood sample) for 15 min at 37 0C. The incubated blood samples were again analyzed for TLC and DLC. Neutrophil index and percent neutrophil adhesion was calculated as follows,
Neutrophil Index (NI) = TLC X Percentageneutrophils.Where,NIu: Neutrophil Index before incubation with nylon fibers.NIt: Neutrophil Index after incubation with nylon fibers.
Cyclophosphamide induced neutropenia
Cyclophosphamide induced neutropenia method was as described by Thatte et al. (1987)24 and Sudha et al. (2010)25 . Swiss albino mice were treated orally for 10 days. On 10th day neutropenic dose of cyclophosphamide (200 mg/kg, p.o) was injected and this day was labeled as day zero. Blood was collected, the total leukocyte count (TLC) and DLC were performed prior to and on day 3 after injection of cyclophosphamide. The TLC and neutrophil counts (%) in treated groups were compared with the values of the control.
Statistical analysis:
The statistical significance was assessed using one-way analysis of variance (ANOVA) followed by Dunnet’s comparison test. The values are expressed as mean ± SEM and P<0.05 was considered significant.
|
Back to Top  |
|
 |
 |
|
Delayed type hypersensitivity (DTH)
In the immune suppressed groups, where the immunity was suppressed by administration of
cyclophosphamide, aqueous extract showed potentiation of DTH response in terms of significant increase in the mean difference in footpad thickness up to 2.72 mm and 2.56 mm at the doses of 50 and 100 mg/kg, respectively (Figure 1).
Neutrophil adhesion test:
Incubation of neutrophils with nylon fibers (NF) produced a decrease in the neutrophil counts due to adhesion of neutrophils to the fibers. All the extracts showed significant increase in the neutrophil adhesion compared to control. Percentage adhesion in aqueous extract, 45.75%and 44.57% at 50 and 100 mg/kg, respectively, is found to better then petroleum ether and ethanol extracts (Figure 2).
Cyclophosphamide induced neutropenia :
Administration of cyclophosphamide reduced the TLC in control animals by 69.6%. Pretreatment of animals with aqueous extract for 10 days before cyclophosphamide administration produced 33.85% and 32.23% reduction in TLC with low and high doses respectively. The pretreatment of animals with petroleum ether and ethanol extract showed 46.23% and 40.32% and 48.17% and 41.02% reduction in TLC with low and high doses respectively compared to initial values. The percentage reduction in neutrophil count was found to be 43.90% in control and 15.60% and 13.20% with low and high doses of aqueous extract respectively (Figure 3).
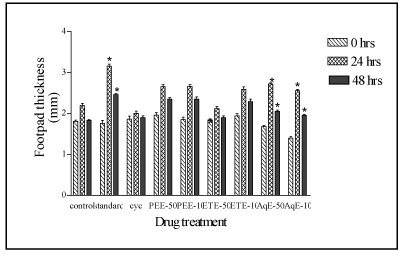
Figure 1. Effect of various extracts of F. racemosa bark on cell-mediated immune response by delayed type hypersensitivity induced footpad edema. Results are expressed as Mean ± S. E. M. (n=6); *=p<0.05 compared with control. ‘cyc’ is negative control group, PEE is petroleum ether extract, ETE is ethanol extract and AqE is aqueous extract.
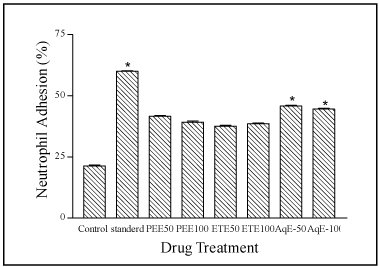
Figure 2. Effect of various extracts of F. racemosa bark on neutrophil adhesion test. Results are expressed as Mean ± S. E. M. (n=6); *=p<0.05 . compared with control group, PEE is petroleum ether extract, ETE is ethanol extract and AqE is Date of manuscript submission:aqueous extract.
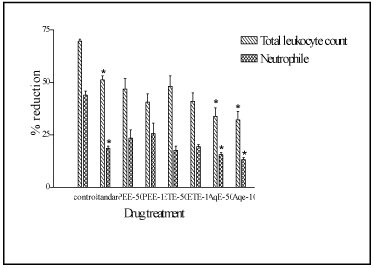
Figure 3. Effect of various extracts of F.
racemosa bark on cyclophosphamide induced
neutropenia
|
Back to Top  |
|
 |
 |
|
Recently phytopharmaceutical research received much attention to develop safe and effective lead compounds with potential immunomodulatory activity. The present study was undertaken to validate folk use of F. racemosa as an immunomodulatory agent. Constant increase in antibiotic resistant strains of microorganisms has prompted scientists to look for herbal immunomodulators to treat various infections. Modulation of immune response through stimulation or suppression may help in maintaining a disease free state26 . Hence in the present study the immunomodulatory activity of petroleum ether, ethanol, and aqueous extracts of bark of this plant was investigated. The DTH response, which is a direct correlate of cell mediated immunity (CMI), was found to be significantly increased at a dose of 50 and 100 mg/kg/day for aqueous extract.
During CMI responses, sensitized T-lymphocytes, when challenged by the antigen, are converted to lymphoblasts and secrete lymphokines, attracting more scavenger cells to the site of reaction. The infiltrating cells are thus immobilized to promote defensive (inflammatory) reaction. In our studies, foot volume was enhanced after aqueous treatment suggesting cell mediated immune enhancement. Increase in the DTH response indicates that aqueous extract has a stimulatory effect on lymphocytes and accessory cell types required for the expression of the reaction27 . Cytokines are secreted by activated immune cells for margination and extravasation of the phagocytes mainly polymorphonuclear neutrophils. Significantly evoked increase in the adhesion of neutrophils to nylon fibers which correlates to the process of margination of cells in blood vessels28. In the present study, aqueous extract evoked a significant increase in percent of neutrophils. This may help in increasing immunity of body against microbial infections. The cyclophosphamide induced neutropenia model concentrates on the protective effects against cyclophosphamide induced myelosuppression in the experimental animals29 . Both low and high doses of aqueous extract caused decrease in the cyclophosphamide induced neutropenia suggesting that it attenuates the effect of cyclophosphamide on the haemopoetic system. It can be concluded that immunostimulant activity of aqueous extract was more pronounced and thus it has great potential as a source for natural health products.
Extrapolation of animal data to a clinical setting is not always appropriate. Nevertheless the interesting results, we obtained, indicate that human studies are needed to evaluate this bark for their immunomodulatory studies. The use of F. racemosa bark as an immunomodulator may be possible in future.
|
Back to Top  |
|
 |
 |
|
1. Sairam M, Sharma SK, Ilavazhagan G, Kumar D & Selvamurthy W, Immunomodulatory effects of NIM-76, a volatile fraction from Neem oil, J Ethnopharmacol 55 (1997) 133.
2. Gabhe SN, Tatke PA & Khan TA, Evaluation of the immunomodulatory activity of methanol extract of Ficus bengalensis root in rats, Indian J Pharmacol 38 (2006) 271.
3. Thatte UM & Dhanukar SA, Ayurveda and contemporary scientific thought, Trends in Pharmacological Sci 7 (1986) 247.
4. Warrier PK, Indian Medicinal Plants (Orient Longman Ltd, Chennai) 1996, 34.
5.Chopra RN, Chopra IC & Varma BS, Supplement to Glossary of Indian Medicinal Plants (CSIR, New Delhi) 1992, 29.
6.Rao CV, Arti R, Verma M, Vijayakumar & Rastogi S, Gastroprotective effect of standardized extract of Ficus glomerata fruit on experimental gastric ulcers in rats, J Ethnopharmacol 115 (2008) 323.
7. Agrawal S & Misra K, Lecoanthocyanins from the bark of Ficus racemosa, Chem Scr 12 (1977) 37.
8. Agarwal YK, Studies on trunk bark of Ficus racemosa, Rocz Chem 51 (1977) 1265.
9. Bhatt K & Agarwal YK, Chemical investigation of the trunk bark from Ficus racemosa, J Indian Chem Soc 50 (1973) 611.
10. Merchant JR, Bakshi VM & Engineer AB, Chemical investigation of the fruits of Ficus glomerata, Indian J Chem 17 (1979) 87.
11. Suresh C, Jawahar L & Sabir M, Chemical examination of the fruits of Ficus glomerata, J Indian Chem Soc 56 (1979) 1269.
12. Rao CV, Arti R, Verma M, Vijayakumar & Rastogi S, Gastroprotective effect of standardized extract of Ficus glomerata fruit on experimental gastric ulcers in rats, J Ethnopharmacol 115 (2008) 323.
13. Devaraj KB, Gowda LR & Prakash V, An unusual thermostable aspartic protease from the latex of Ficus racemosa, Phytochemistry 69 (2008) 647.
14. Rao BR, Murugesan T, Sinha S, Saha BP, Pal M & Mandal SC, Glucose lowering efficacy of Ficus racemosa barks extract in normal and alloxan diabetic rats, Phytother Res 16 (2002) 590. 1.
15. Ratnasooriya WD, Jayakody JR & Nadarajah T, Antidiuretic activity of aqueous bark extract of Sri Lankan Ficus racemosa in rats, Acta Biol Hung 54 (2003) 357.
16. Biswas TK & Mukerjee B, Plant medicines of Indian origin for wound healing activity–A review, Int J Low Extreme Wounds 2 (2003) 25.
17. Faiyaz A & Asna U, Hepatoprotective effects of Ficus racemosa stem bark against carbon tetrachloride-induced hepatic damage in albino rats, Pharmaceutical Biology 48 (2010) 210.
18. Malairajan P, Geetha GK, Narasimhan S & Jessi KV, Analgesic activity of some Indian Medical plants, J Ethnopharmacol 106 (2006) 425.
19. Fulzele SV, Bhurchandi PM, Kanoje VM, Joshi SB & Dorle AK, Immunostimulant activity of Ashtamangal ghrita in rats, Indian J Pharmacol 34 (2002) 194.
20.Bafna AR & Mishra SH, Immunomodulatory activity of menthol extract of flower‐heads of Sphaeranthus indicus Linn, Ars Pharmaceutica 45 (2004) 281.
21.Devhare SV, Nirmal SA, Rub RA, Dhasade VV, Zaware BB & Mandal SC, Immunomodulating activity of Celosia argentea Linn aerial parts, Lat Am J Pharm 30 (2011) 168.
22.Puri A, Saxen RP, Saxena KC, Srivastava V & Tandon S, Immunostimulant activity of Nyctanthes arbor -tristis L. J Ethnopharmacol 42 (1994) 31.
24.Fulzele SV, Satturwar PM, Joshi & Dorle AK, Studies on the immunomodulatory activity of an herbal formulation, Indian Drugs 39 (2002) 364.
25.Sudha P, Syed MB, Dhamingi SS & 4 Gowda KC, Immunomodulatory activity of methanolic leaf extract of Moringa oleifera in animals, Indian J Physiol Pharmacol 54 (2010) 133.
26.Bafna AR & Mishra SH,Immunomodulatory activity of methanolextract of roots of Cissampelos pareira Linn
|
Back to Top  |
|
 |
|